Innovative Vaccine Strategy Shows Promise
-
by Anoop Singh
- 5
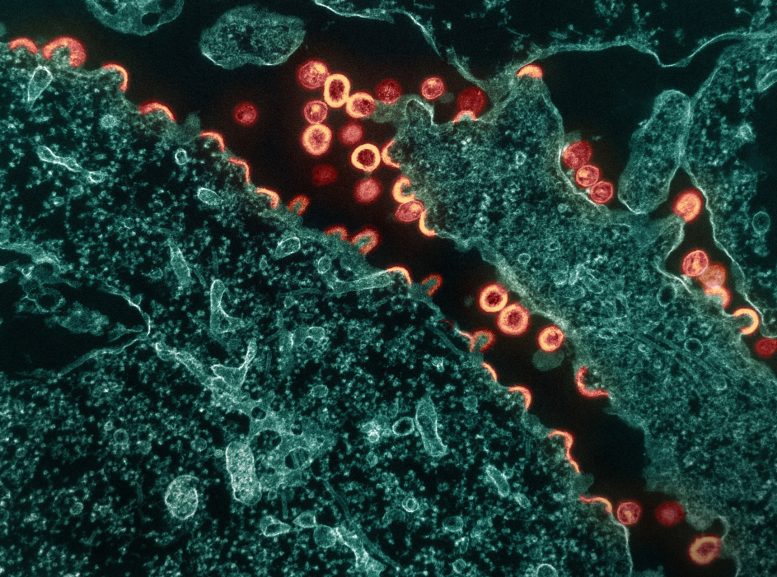
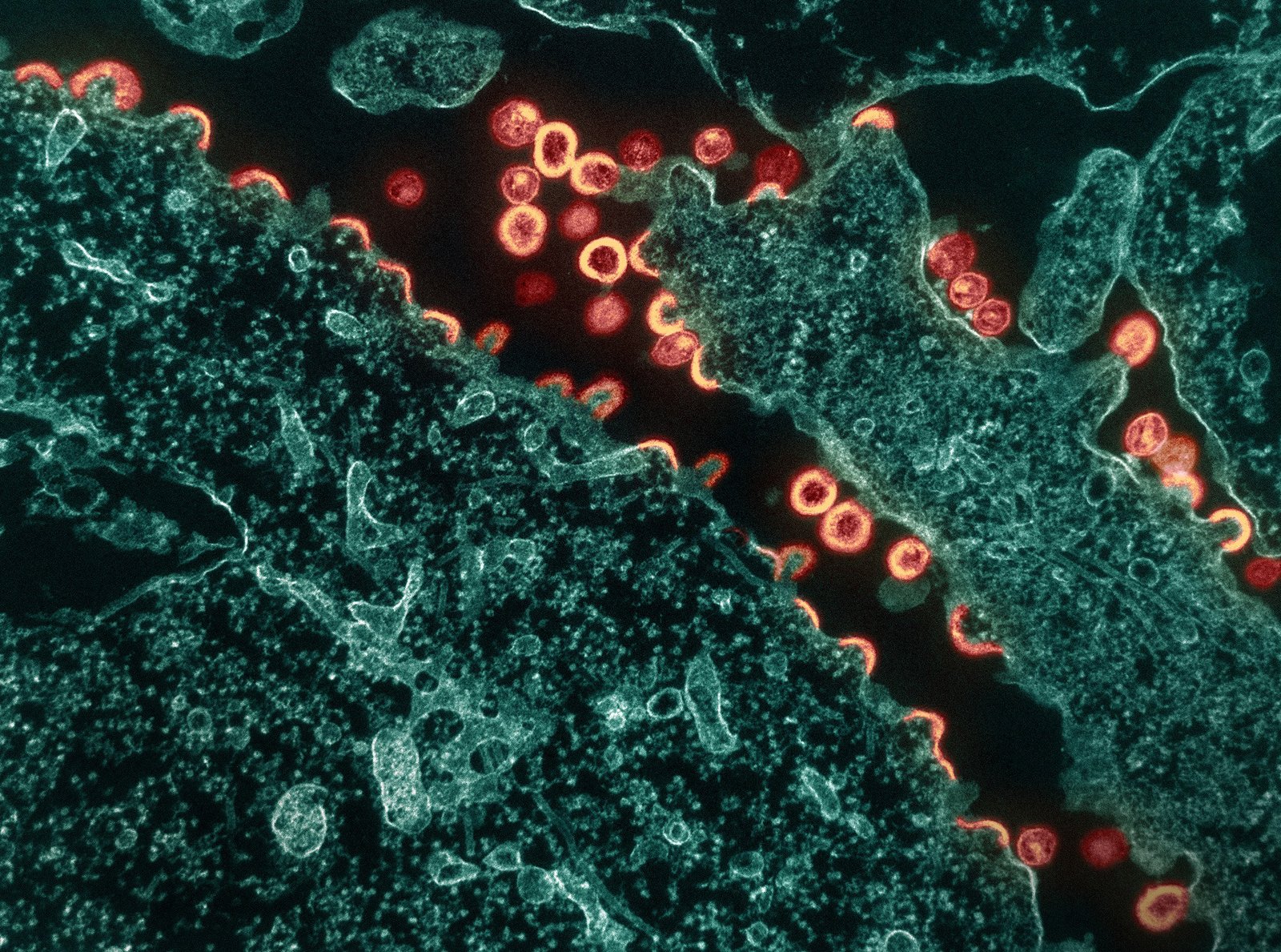
Transmission electron micrograph of HIV-1 virus particles (pink/tan) budding and replicating from a segment of a chronically infected H9 cell (teal). Particles are in various stages of maturity; arc/semi-circles are immature particles that have started to form but are still part of the cell. Immature particles slowly change morphology into mature forms and exhibit the classic “conical or spherical-shaped core.” Image captured at the NIAID Integrated Research Facility (IRF) in Fort Detrick, Maryland. Credit: NIAID
Results from NIH-funded animal models will inform the development of vaccines for humans.
Using advanced immunological techniques, researchers have effectively activated the immune systems of animals to produce rare precursor B cells that can generate a type of HIV broadly neutralizing antibodies (bNAbs). The results, published in Nature Immunology, represent a promising, gradual advance toward creating a preventive HIV vaccine.
HIV is genetically diverse making the virus difficult to target with a vaccine, but bNAbs may overcome that hurdle because they bind to parts of the virus that remain constant even when it mutates. Germline targeting is an immune system-stimulating approach that guides naïve (precursor) B cells to develop into mature B cells that can produce bNAbs. A class of bNAbs called 10E8 is a priority for HIV vaccine development because it neutralizes a particularly broad range of HIV variants.
The 10E8 bNAb binds to a conserved region of the glycoprotein gp41 on HIV’s surface involved in its entry into human immune cells. Designing an immunogen—a molecule used in a vaccine that elicits a specific immune system response—to stimulate the production of 10E8 bNAbs has been challenging because that key region of gp41 is hidden in a recessed crevice on HIV’s surface. Prior vaccine immunogens have not generated bNAbs with the physical structure to reach and bind to gp41.
Advances in Immunogen Design
To address this challenge, the researchers engineered immunogens on nanoparticles that mimic the appearance of a specific part of gp41. They vaccinated rhesus macaque monkeys and mice with those immunogens and elicited specific responses from the 10E8 B cell precursors and induced antibodies that showed signs of maturing into bNAbs that could reach the hidden gp41 region. They observed similar responses when they used mRNA-encoded nanoparticles in mice. The researchers also found that the same immunogens produced B cells that could mature to produce an additional type of gp41-directed bNAb called LN01.
Finally, their laboratory analysis of human blood samples found that 10E8-class bNAb precursors occurred naturally in people without HIV, and that their immunogens bound to and isolated naïve human B cells with 10E8-like features. Together these observations suggest that the promising immunization data from mice and macaques has the potential for translation to humans.
Reference: “Vaccination induces broadly neutralizing antibody precursors to HIV gp41” by Torben Schiffner, Ivy Phung, Rashmi Ray, Adriana Irimia, Ming Tian, Olivia Swanson, Jeong Hyun Lee, Chang-Chun D. Lee, Ester Marina-Zárate, So Yeon Cho, Jiachen Huang, Gabriel Ozorowski, Patrick D. Skog, Andreia M. Serra, Kimmo Rantalainen, Joel D. Allen, Sabyasachi Baboo, Oscar L. Rodriguez, Sunny Himansu, Jianfu Zhou, Jonathan Hurtado, Claudia T. Flynn, Katherine McKenney, Colin Havenar-Daughton, Swati Saha, Kaitlyn Shields, Steven Schulze, Melissa L. Smith, Chi-Hui Liang, Laura Toy, Simone Pecetta, Ying-Cing Lin, Jordan R. Willis, Fabian Sesterhenn, Daniel W. Kulp, Xiaozhen Hu, Christopher A. Cottrell, Xiaoya Zhou, Jennifer Ruiz, Xuesong Wang, Usha Nair, Kathrin H. Kirsch, Hwei-Ling Cheng, Jillian Davis, Oleksandr Kalyuzhniy, Alessia Liguori, Jolene K. Diedrich, Julia T. Ngo, Vanessa Lewis, Nicole Phelps, Ryan D. Tingle, Skye Spencer, Erik Georgeson, Yumiko Adachi, Michael Kubitz, Saman Eskandarzadeh, Marc A. Elsliger, Rama R. Amara, Elise Landais, Bryan Briney, Dennis R. Burton, Diane G. Carnathan, Guido Silvestri, Corey T. Watson, John R. Yates III, James C. Paulson, Max Crispin, Gevorg Grigoryan, Andrew B. Ward, Devin Sok, Frederick W. Alt, Ian A. Wilson, Facundo D. Batista, Shane Crotty and William R. Schief, 30 May 2024, Nature Immunology.
DOI: 10.1038/s41590-024-01833-w
The research was conducted by the Scripps Consortium for HIV/AIDS Vaccine Development, one of two consortia supported by the National Institutes of Health’s (NIH) National Institute of Allergy and Infectious Diseases (NIAID). The research also was supported by collaborating partners including the Bill & Melinda Gates Foundation and other NIH Institutes and Offices. According to the authors, these findings support the development of the immunogens as the first part of a multi-step vaccine regimen for humans. Their work further supports research in developing a germline-targeting strategy for priming the immune system to elicit a bNAb called VRC01. This bNAb was discovered by NIAID researchers almost 15 years ago. The goal of this line of research is to develop an HIV vaccine that generates multiple classes of bNAbs to prevent HIV.
Transmission electron micrograph of HIV-1 virus particles (pink/tan) budding and replicating from a segment of a chronically infected H9 cell (teal). Particles are in various stages of maturity; arc/semi-circles are immature particles that have started to form but are still part of the cell. Immature particles slowly change morphology into mature forms and exhibit the…
Transmission electron micrograph of HIV-1 virus particles (pink/tan) budding and replicating from a segment of a chronically infected H9 cell (teal). Particles are in various stages of maturity; arc/semi-circles are immature particles that have started to form but are still part of the cell. Immature particles slowly change morphology into mature forms and exhibit the…
