
Boronic Acid-Powered Enzyme Yields Groundbreaking Catalysis
-
by Anoop Singh
- 22

Scientists have created an enzyme that incorporates boronic acid, which enhances its catalytic power and selectivity through directed evolution, offering a more sustainable alternative to traditional chemical reactions. This innovation not only speeds up catalysis but also achieves high enantioselectivity, crucial for producing chiral molecules needed in pharmaceuticals.
Boronic acid has been utilized in organic chemistry for decades, despite not being found in any living organism.
‘It gives rise to different chemical reactions than those we find in nature,’ explains Gerard Roelfes, Professor of Biomolecular Chemistry & Catalysis at the University of Groningen. His group created an enzyme with boronic acid at its reactive center and then used directed evolution to make it more selective and to improve its catalytic power.

This is Gerard Roelfes, Professor of Biomolecular Chemistry & Catalysis at the University of Groningen and lead author of the Nature paper. Credit: University of Groningen
Furthermore, enzymatic reactions are more sustainable than classical chemical reactions, as they take place at low temperatures and without toxic solvents. The study was presented online in the journal Nature on 8 May.
The application of boron in organic chemistry dates back some seventy years and was awarded a Nobel Prize for Chemistry in 1979. In recent years, the interest in boron as a catalyst has grown, but as yet, its use in the chemical industry is limited. Roelfes: ‘So far, boron catalysis is too slow and it is not very suitable for enantioselective reactions.’
These types of reactions are used to create chiral molecules, which can exist in two versions that are mirror images of each other, like a left and a right hand. In many drugs, both ‘hands’ can have a different effect. It is, therefore, important to selectively produce the proper ‘hand’, especially for the pharmaceutical industry.
Expanded genetic code
‘To make this possible, we set out to introduce boron into an enzyme. Our group has a long history of designing enzymes that don’t exist in nature.’ The Roelfes group used an expanded genetic code to introduce a non-natural amino acid that contains a reactive boronic acid group into an enzyme. ‘Using this technique, we can determine at the DNA level where we place the amino acid in a protein.’
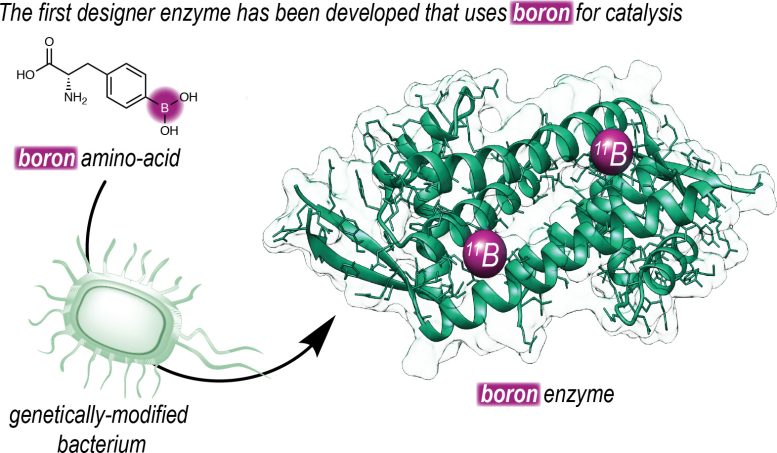
This is a schematic representation of how University of Groningen chemists introduced an artificial amino acid containing boron into an enzyme. Credit: Roefels Lab / University of Groningen
Once they made an enzyme with boronic acid at its reactive center, they could use directed evolution to increase its efficiency, resulting in faster catalysis. ‘Furthermore, by placing the boronic acid in the chiral context of an enzyme, we were able to achieve highly enantioselective catalysis.’ The reaction that is described in the journal Nature is a ‘proof of principle’ and shows the way to harnessing the catalytic power of boron in enzymes.
Biocatalysis
Using enzymes to create organic compounds is important for the pharmaceutical industry.
‘In their push towards greener and more sustainable ways of producing drugs, they are looking at biocatalysis to replace conventional chemical reactions.’ At the University of Groningen, concerted efforts are being made towards this goal. ‘We have a number of research groups at the Faculty of Science and Engineering engaged in this kind of work, using different approaches to create biocatalytic solutions for the chemical industry.’ In this context, Roelfes and his team will continue to develop their boronic acid enzymes and create other such new-to-nature enzymes.
Reference: “Boron catalysis in a designer enzyme” by Lars Longwitz, Reuben B. Leveson-Gower, Henriëtte J. Rozeboom, Andy-Mark W. H. Thunnissen and Gerard Roelfes, 8 May 2024, Nature.
DOI: 10.1038/s41586-024-07391-3
Scientists have created an enzyme that incorporates boronic acid, which enhances its catalytic power and selectivity through directed evolution, offering a more sustainable alternative to traditional chemical reactions. This innovation not only speeds up catalysis but also achieves high enantioselectivity, crucial for producing chiral molecules needed in pharmaceuticals. Boronic acid has been utilized in organic…
Scientists have created an enzyme that incorporates boronic acid, which enhances its catalytic power and selectivity through directed evolution, offering a more sustainable alternative to traditional chemical reactions. This innovation not only speeds up catalysis but also achieves high enantioselectivity, crucial for producing chiral molecules needed in pharmaceuticals. Boronic acid has been utilized in organic…
